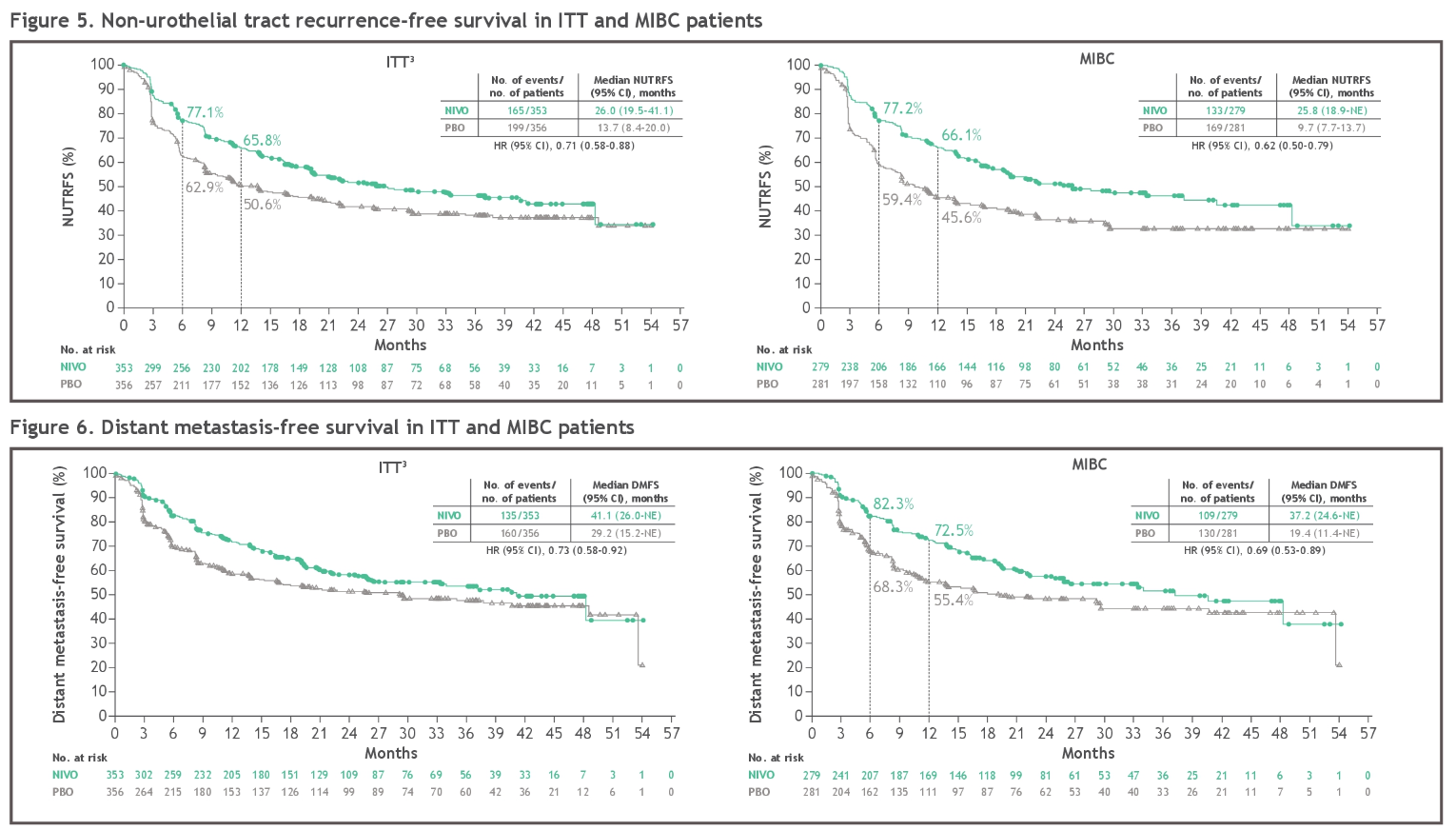 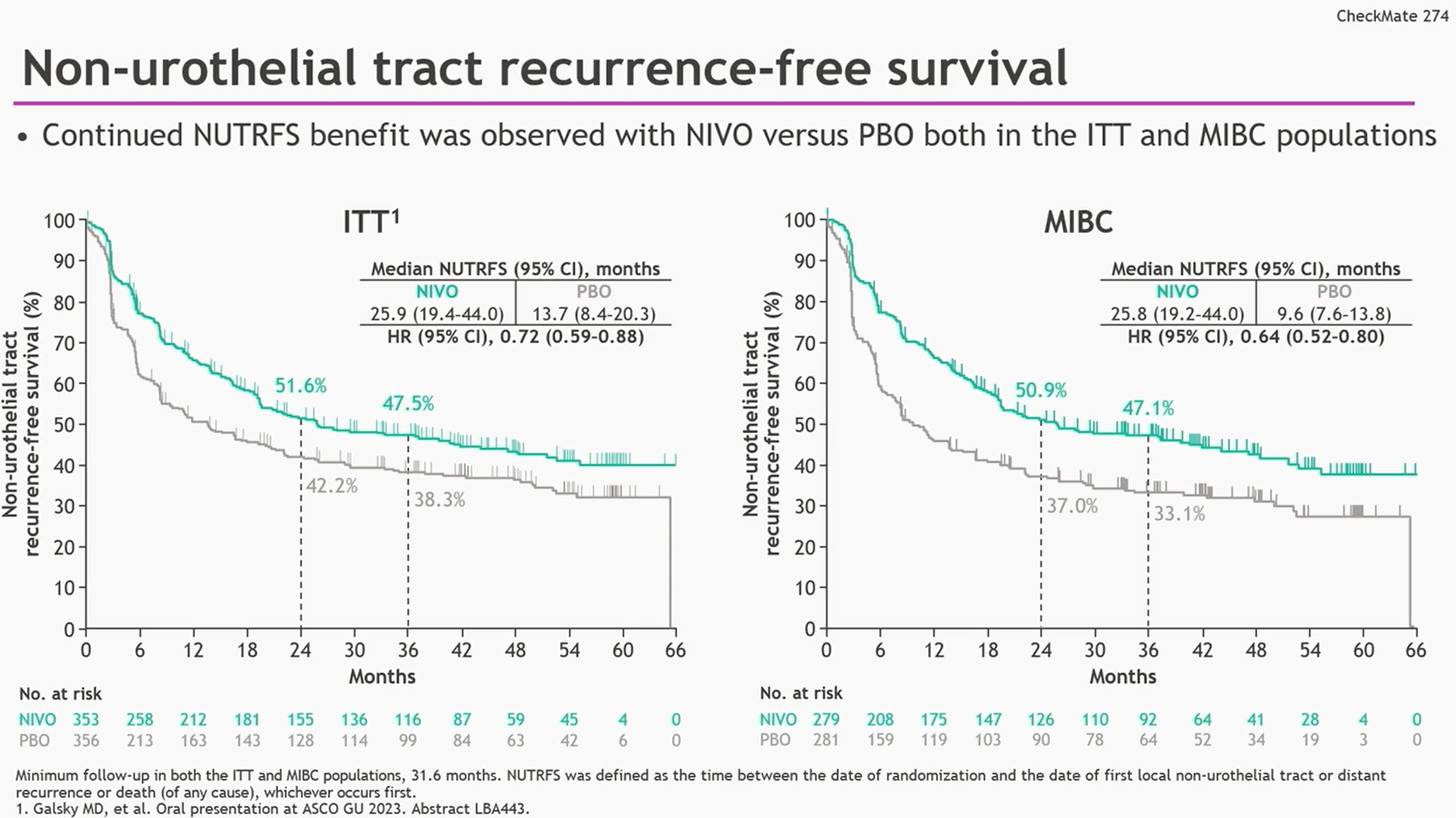
DFS results with nivolumab were consistent across all pre-specified subgroups, except for two very small subsets of patients with renal pelvis and ureter carcinoma. At a median follow-up of 36.1 months, the primary endpoint of DFS was doubled with nivolumab in the intent-to-treat (ITT) population (22.0 months vs 10.9 months with placebo HR: 0.71 ), with considerably pronounced DFS improvement in the programmed death-ligand 1 (PD-L1) ≥1% population (52.6 months vs 8.4 months HR: 0.52 ). 4 Patients were randomized 1:1 to receive either nivolumab (240 mg) (n=353) or placebo (n=356) every 2 weeks for up to a year. 4īriefly, the trial enrolled patients with ypT2-ypT4a or ypN+ MIUC who have had neoadjuvant cisplatin chemotherapy and patients with pT3-pT4a or pN+ MIUC who did not have prior neoadjuvant cisplatin chemotherapy and were not eligible for or refused cisplatin chemotherapy. Updated results with a minimum follow-up of 31.6 months presented at this year’s ASCO GU 2023 corroborated the clinical benefit of nivolumab in MIUC patients. Results from the phase III, placebo-controlled CheckMate 274 trial, investigating adjuvant nivolumab in patients with HR muscle-invasive urothelial carcinoma (MIUC), 3 ultimately led to the regulatory approval of nivolumab for use in this clinical setting. CheckMate 274: Adjuvant nivolumab improves disease-free survival in patients with high-risk MIUC 2 Overall, these results support the use of pembrolizumab in patients with papillary HR NMIBC unresponsive to BCG. These also included patients with persistent and recurrent disease status at baseline, but not for the subset of patients with progressive disease. This DFS benefit was uniform across nearly all prespecified subgroups based on demographic characteristics and tumor patterns. 2 At 24- and 36-month landmark analyses, the DFS rate was sustained at 34.9%. The primary endpoint of the 12-month disease-free survival (DFS) rate was 43.5%. The overall median follow-up was 45.4 months. At 24 weeks, patients showing no recurrence or progression of the disease continued to receive pembrolizumab for up to 2 years otherwise, treatment was discontinued. 2 After 12 weeks of therapy, patients who did not achieve complete response (CR) discontinued the treatment. 1 In this ongoing study, pembrolizumab monotherapy was also assessed in 132 BCG-unresponsive patients with papillary tumors (high-grade Ta or any T1) without CIS (cohort B). Previous findings from the phase II KEYNOTE-057 trial showed that pembrolizumab is tolerable and invokes a promising anti-tumor activity in patients with high-risk (HR) non-muscle invasive bladder cancer (NMIBC) unresponsive to bacillus Calmette–Guérin (BCG), with carcinoma in situ (CIS) (cohort A). KEYNOTE-057: Pembrolizumab shows clinical benefit in patients with high-risk non-muscle invasive bladder cancer
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
